Introduction to Photoionization
A Photo-Ionization Detector (PID) measures real-time concentrations of organic and inorganic vapors in low levels from parts-per-billion (ppb) up to 10,000 parts-per-million (ppm). A PID is a very sensitive instrument used in a wide variety of applications such as:
- Hazardous waste sites
- Clean-up of spills
- Protecting workers from toxic vapors
- Refineries
- Leak detection for compliance
- Pulp and paper plants
- Hazardous materials response
- Terrorist chemical attacks
How Does a PID Work?
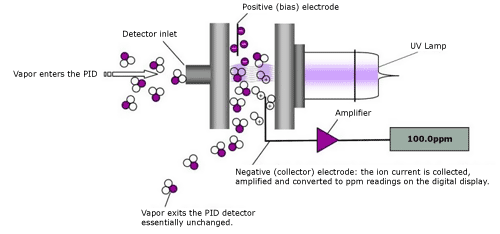
A PID uses an ultraviolet (UV) lamp to ionize molecules, causing them to release an electron and form a positive ion. This will occur when the Ionization Potential (IP) of the molecule is less than the electron volt (eV) output of the UV lamp (i.e., 10.6eV, 11.7eV, or 11.8eV). Ions formed in the air are driven in one direction by a bias electrode and accumulated at a collecting electrode. The ion current is then amplified and converted to a digital meter readout in parts-per-million (ppm). The most common UV lamps used in field work are 10.6eV, 11.7eV, and 11.8eV. The useful lifespan of the 10.6EV is about 3,000 hours, the 11.7eV is about 200 hours, and the 11.8eV is about 500 hours. Of course, these lifespans are all dependent on how carefully the lamps are used and maintained.
Photo-Ionization Detector (PID)