X-RAY FLUORESCENCE (XRF) SPECTROMETRY
Basic Theory
Although more popularly known for its diagnostic use in the medical field, the use of x-rays forms the basis of many other powerful measurement techniques, including X-ray Fluorescence (XRF) Spectrometry.
XRF Spectrometry is used to identify elements in a substance and quantify the amount of those elements present to ultimately determine the elemental composition of a material. An element is identified by its characteristic X-ray emission wavelength ( λ ) or energy (E). The amount of an element present is quantified by measuring the intensity (I) of its characteristic emission.
All atoms have a fixed number of electrons (negatively charged particles) arranged in orbitals around the nucleus. Energy Dispersive (ED) XRF and Wavelength Dispersive (WD) XRF Spectrometry typically utilize activity in the first three electron orbitals, the K, L, and M lines, where K is closest to the nucleus.
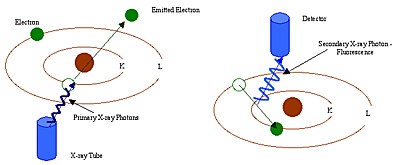
In XRF Spectrometry, high-energy primary X-ray photons are emitted from a source (X-ray tube) and strike the sample. The primary photons from the X-ray tube have enough energy to knock electrons out of the innermost, K or L, orbitals. When this occurs, the atoms become ions, which are unstable. An electron from an outer orbital, L or M, will move into the newly vacant space at the inner orbital to regain stability. As the electron from the outer orbital moves into the inner orbital space, it emits an energy known as a secondary X-ray photon. This phenomenon is called fluorescence. The secondary X-ray produced is characteristic of a specific element. The energy (E) of the emitted fluorescent X-ray photon is determined by the difference in energies between the initial and final orbitals of the individual transitions.
This is described by the formula
![]()
where h is Planck's constant; c is the velocity of light; and λ is the characteristic wavelength of the photon.
Energies are inversely proportional to the wavelengths; they are characteristic for each element. For example the Kα energy for Iron (Fe) is about 6.4keV. Typical spectra for EDXRF Spectrometry appear as a plot of Energy (E) versus the Intensity (I).
Elemental Analysis
XRF Spectrometry is the choice of many analysts for elemental analysis. XRF Spectrometry easily and quickly identifies and quantifies elements over a wide dynamic concentration range, from PPM levels up to virtually 100% by weight. XRF Spectrometry does not destroy the sample and requires little, if any, sample preparation. It has a very fast overall analysis turnaround time. These factors lead to a significant reduction in the per sample analytical cost when compared to other elemental analysis techniques.
Aqueous elemental analysis instrument techniques typically require destructive and time-consuming specimen preparation, often using concentrated acids or other hazardous materials. Not only is the sample destroyed, waste streams are generated during the analysis process that need to be disposed of, many of which are hazardous. These aqueous elemental analysis techniques often take twenty minutes to several hours for sample preparation and analysis time. All of these factors lead to a relatively high cost per sample. However, if PPB and lower elemental concentrations are the primary measurement need, aqueous instrument elemental analysis techniques are necessary.
All elemental analysis techniques experience interferences, both chemical and physical in nature, and must be corrected or compensated for in order to achieve adequate analytical results. Most aqueous instrument techniques for elemental analysis suffer from interferences that are corrected for by extensive and complex sample preparation techniques, instrumentation modifications or enhancements, and by mathematical corrections in the system's software. In XRF Spectrometry, the primary interference is from other specific elements in a substance that can influence (matrix effects) the analysis of the element(s) of interest. However, these interferences are well known and documented; and, instrumentation advancements and mathematical corrections in the system's software easily and quickly correct for them. In certain cases, the geometry of the sample can affect XRF analysis, but this is easily compensated for by selecting the optimum sampling area, grinding or polishing the sample, or by pressing a pellet or making glass beads.
Quantitative elemental analysis for XRF Spectrometry is typically performed using Empirical Methods (calibration curves using standards similar in property to the unknown) or Fundamental Parameters (FP). FP is frequently preferred because it allows elemental analysis to be performed without standards or calibration curves. This enables the analyst to use the system immediately, without having to spend additional time setting up individual calibration curves for the various elements and materials of interest. The capabilities of modern computers allow the use of this no-standard mathematical analysis, FP, accompanied by stored libraries of known materials, to determine not only the elemental composition of an unknown material quickly and easily, but even to identify the unknown material itself.
Spectrometers
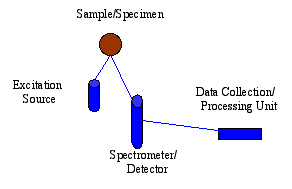
Innov-X Systems utilizes the EDXRF Spectrometer technique for its mechanical simplicity and excellent adaptation to portable field use. An EDXRF system typically has three major components: an excitation source, a spectrometer/detector, and a data collection/processing unit. The ease of use, rapid analysis time, lower initial purchase price and substantially lower long-term maintenance costs of EDXRF Spectrometers have led to having more systems in use today worldwide than WDXRF Spectrometer systems. Handheld, field portable EDXRF units can be taken directly to the sample as opposed to bringing the sample to the analyzer and configuring it to fit in an analysis chamber. Innov-X Systems portable, handheld EDXRF units solve real 21 st century application problems: solving crimes, analyzing alloys, exposing pollution, preserving history, searching for WMD's, conserving art treasures, and a myriad of other elemental field-oriented analyses.